Hepatocellular carcinoma (HCC), the most common 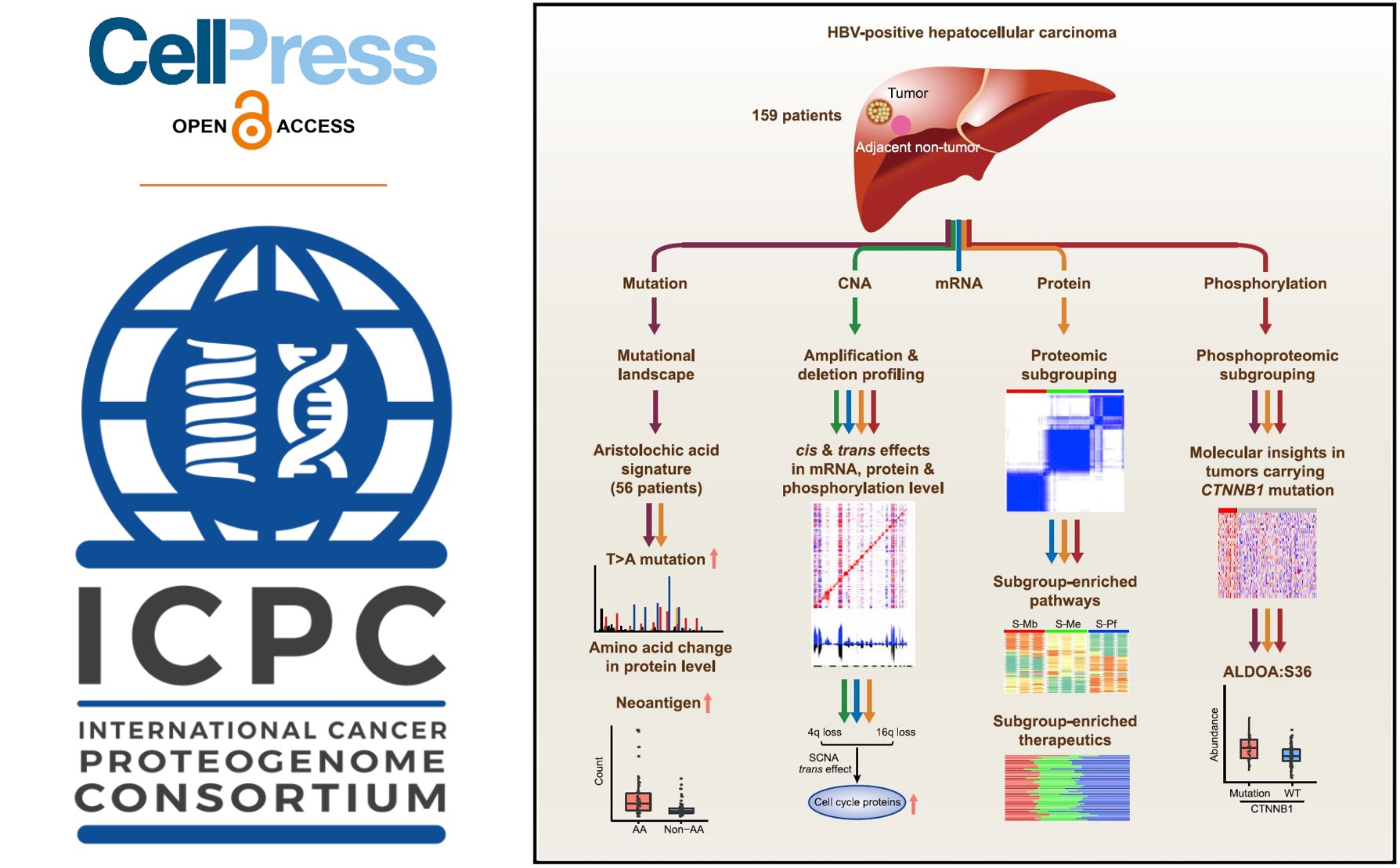 form of liver cancer, occurs most often in people with hepatitis B or C viral (HBV or HCV) infections who drink large amounts of alcohol. Unlike the recent progress of antiviral-eliminating therapy for HCV, current antiviral therapy is only able to reduce rather than eliminate HBV and is estimated to affect 292 million people globally. It is estimated that HBV-related HCC accounts for 85% of HCC cases in China due to the prevalence of HBV infection. Therefore, there is a need to understand how genetic changes drive cancer phenotype in HBV-related HCC.
form of liver cancer, occurs most often in people with hepatitis B or C viral (HBV or HCV) infections who drink large amounts of alcohol. Unlike the recent progress of antiviral-eliminating therapy for HCV, current antiviral therapy is only able to reduce rather than eliminate HBV and is estimated to affect 292 million people globally. It is estimated that HBV-related HCC accounts for 85% of HCC cases in China due to the prevalence of HBV infection. Therefore, there is a need to understand how genetic changes drive cancer phenotype in HBV-related HCC.
The new study, a comprehensive multi-omics analysis of paired tumor and adjacent non-tumor liver tissues from a Chinese cohort of 159 patients by the ICPC teams from China, found oncogenes, tumor suppressors, activation of key signaling pathways, and liver-specific metabolic reprogramming of HBV-related HCC. Leveraging CPTAC pipelines, investigators analyzed the samples by integrating mass spectrometry (MS)-based proteomic, phosphoproteomics, transcriptomic profiling, and whole exome sequencing. The study appears online October 3rd, 2019, in Cell.
Proteomic profiling identified three subgroups associated with clinical and molecular attributes including patient survival, tumor thrombus, genetic profile, and correlation with the liver-specific proteome. "These proteomic subgroups have distinct features in metabolic reprogramming, microenvironment dysregulation, cell proliferation, and subsequent potential therapeutics," says study co-author Hu Zhou, a proteomic scientist at Shanghai Institute of Materia Medica, Chinese Academy of Sciences.
Investigators also identified PYCR2, an enzyme involved in amino acid production, and ADH1A, an alcohol metabolizing enzyme, as prognostic protein biomarkers in the Chinese HCC-HBV cohort. They hypothesized that the gain-of-function for PYCR2 may enable tumor cells with an extra capability to cope with excessive oxidative species from enhanced metabolism while ADH1A (down-regulated in tumor tissues) impairs normal metabolic function of the liver.
Additionally, investigators looked at the effects of aristolochic acid (AA) and their derivatives, which are part of traditional Chinese medicine and used as an herbal extract to treat HCC. Recent evidence suggests that AA and its derivatives may contribute to HBV-related HCC. But until now, there has been no evidence that AA- signatures due to genetic mutations could be translated and detected at the protein level. In the Chinese HCC-HBV cohort, investigators identified 56 cases with AA-signature and detected two peptides encoded by mutated genes with AA-signature mutations. The result indicates that herbal medicine as a treatment option could result in mutated protein products in addition to affecting genome stability. Additionally, the finding suggests that even herbal medicines containing AA or their derivatives may also have an effect in HBV-related HCC.
This study not only generated high quality data, which has limited access through the Chinese database National Omics Data Encyclopedia (NODE) or open access through the CPTAC Data Portal (soon-to-be available), but promotes hypothesis-driven questions and provides biological insights underlying clinical features of HCC. This work was done under the auspices of a Memorandum of Understanding between the Shanghai Institute of Materia Medica of the Chinese Academy of Sciences, Fudan University, and the U.S. National Cancer Institute’s Office of Cancer Clinical Proteomics Research (OCCPR) through the Clinical Proteomic Tumor Analysis Consortium (CPTAC) and inspired by the NCI Cancer MoonshotSM. CPTAC collaborates with international organizations/institutions to accelerate the understanding of the molecular basis of cancer through the application of proteogenomics, standards development, and publicly available datasets.

